Increasing the mole fraction of the lighter element (P) results in a larger band gap, and thus a higher energy of emitted photons. For example, red and orange light-emitting diodes (LED's) are made from solid solutions with compositions of GaP 0.40As 0.60 and GaP 0.65As 0.35, respectively. This "law" is often violated in real materials, but nevertheless offers useful guidance for designing materials with specific band gaps. Often, there is a linear relation between composition and band gap, which is referred to as Vegard's Law. Semiconductor solid solutions such as GaAs 1-xP x have band gaps that are intermediate between the end member compounds, in this case GaAs and GaP (both zincblende structure). 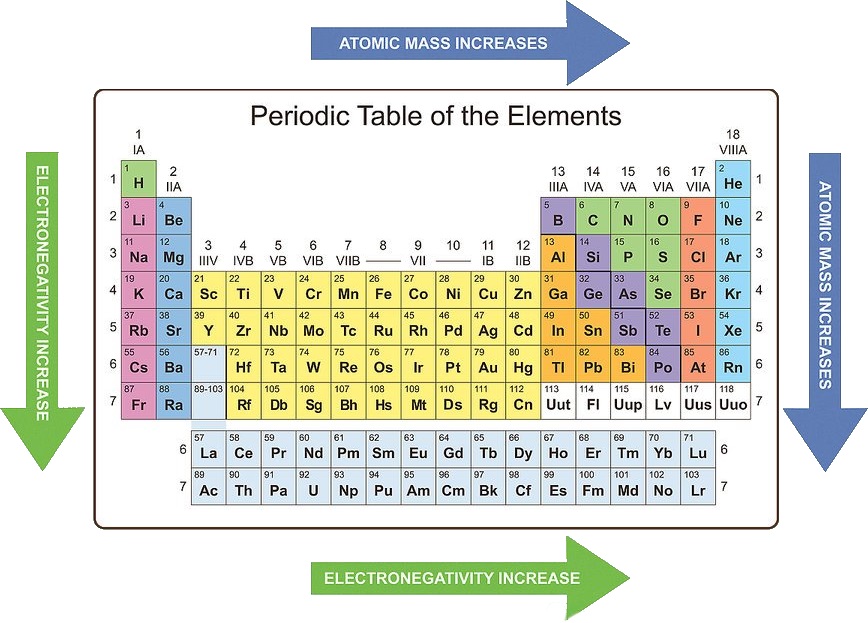
Wider gap materials (Si, GaAs, GaP, GaN, CdTe, CuIn xGa 1-xSe 2) are used in electronics, light-emitting diodes, and solar cells.Ĭolor wheel showing the colors and wavelengths of emitted light.Narrow gap materials (Hg xCd 1 - xTe, VO 2, InSb, Bi 2Te 3) are used as infrared photodetectors and thermoelectrics (which convert heat to electricity).Many of the applications of semiconductors are related to band gaps: The band gap is a very important property of a semiconductor because it determines its color and conductivity. As the electronegativity difference Δχ increases, so does the energy difference between bonding and antibonding orbitals. This trend can also be understood from a simple MO picture, as we discussed in Ch. (2) For isoelectronic compounds, increasing ionicity results in a larger band gap. This difference decreases (and bonds become weaker) as the principal quantum number increases. This trend can be understood by recalling that E gap is related to the energy splitting between bonding and antibonding orbitals. Semiconductors, as we noted above, are somewhat arbitrarily defined as insulators with band gap energy Si > Ge > α-Sn 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
